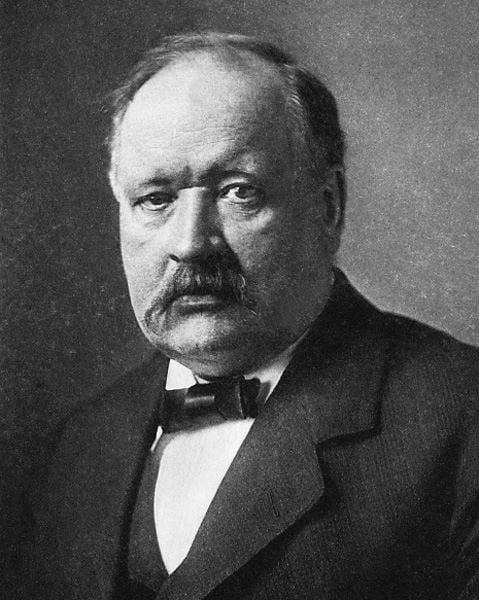Who shared the early development of physical chemistry with Ostwald?
Albert Einstein
Wilhelm Roentgen
Svante Arrhenius
Linus Pauling
Answer
Arrhenius, a Swedish chemist, played a pivotal role in the early development of physical chemistry alongside Ostwald. His contributions revolve around the revolutionary theory of electrolytic dissociation, which elucidates that when an electrolyte dissolves in a solvent, it breaks up into positively charged ions (cations) and negatively charged ions (anions). This concept revolutionized our understanding of chemical reactions in solutions and laid the foundation for modern electrochemistry.

Unraveling the Legacy: The Wilhelm Ostwald Challenge
Related Quizzes

Subscribe Now!
Learn something
new everyday
Playing quizzes is free! We send 1 quiz every week to your inbox