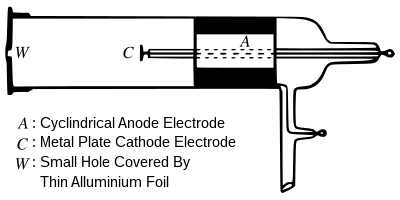
The ejected electrons from a cathode depend on what?
The temperature
The intensity of light
The wavelength of light
The pressure
Answer
The emission of electrons from a cathode, known as the photoelectric effect, depends on the wavelength of incident light. Shorter wavelengths or higher frequencies correspond to more energetic photons carrying enough energy to overcome the material's binding energy and release electrons. This fundamental relationship challenges classical physics, leading to significant breakthroughs in quantum mechanics and shaping our understanding of light-matter interactions at the microscopic level.

Exploring the Scientific Laurels of Philipp Lenard: Uncover the Delights of a Hungarian-German Physicist!
Related Quizzes

Subscribe Now!
Learn something
new everyday
Playing quizzes is free! We send 1 quiz every week to your inbox